Encryption is everywhere in connected medical devices. Data in transit uses TLS. Firmware updates rely on signed packages. Mobile apps pair over Bluetooth. Cloud services authenticate device identity. All of it depends on one fragile foundation: how cryptographic keys are exchanged, provisioned, and managed.
This guide explains key exchange through a medical device cybersecurity lens. You’ll learn where key exchange breaks in the real world, what protocols and patterns reduce risk, and what evidence is worth documenting for FDA-facing submissions and lifecycle security.
Related: If your team is building (or packaging) cryptography evidence for FDA submissions, see our FDA premarket cybersecurity services.
Why key exchange matters in medical device cybersecurity
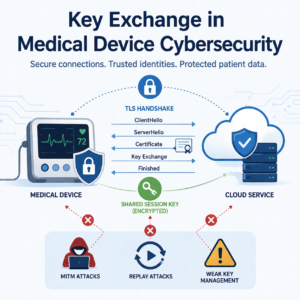
Key exchange is the process of establishing shared secrets (or trusted public keys) so two endpoints can communicate securely. In medical devices, key exchange failures rarely look like “encryption is broken.” They look like:
- A device accepts a fake cloud endpoint because certificate validation was weak.
- A service tool can downgrade the protocol and replay a prior session.
- A BLE pairing flow leaks keys, enabling unauthorized control or data access.
- An update mechanism trusts the wrong signing key, or cannot recover from key compromise.
From a patient safety perspective, these issues can turn into loss of availability (device disruption), loss of integrity (tampered therapy settings), or loss of confidentiality (PHI exposure). That is why modern guidance treats cybersecurity as part of safety and effectiveness, across the total product lifecycle.
Where key exchange shows up in medical devices
You typically see key exchange in four places:
- Device to cloud: TLS sessions, mutual authentication, API tokens and refresh flows.
- Device to mobile app: pairing, onboarding, and session key negotiation (often BLE).
- Device to device: interoperability channels inside a system-of-systems.
- Updates and maintenance: firmware signing keys, secure download, and verification.
Blue Goat Resources:
- TLS 1.3 for medical devices
- Symmetric encryption and key management in devices
- Key escrow tradeoffs for regulated products
The key exchange problem (in plain language)
The “key exchange problem” is simple: how do two parties create a shared secret over an untrusted network without an attacker learning or manipulating it?
If you just “send the key,” you lose. If you exchange keys without authenticating identities, you invite man-in-the-middle attacks. If you reuse keys too broadly, you increase blast radius and reduce forward secrecy.
Modern key establishment schemes (like Diffie-Hellman variants) are designed to solve the shared-secret portion of this problem, but they still require correct authentication and correct implementation choices.
Common key exchange failures that create FDA-worthy risk
1) Man-in-the-middle (MitM) during onboarding or TLS setup
A MitM attack happens when an attacker positions themselves between endpoints and impersonates each side. In devices, MitM risk spikes during:
- first-use pairing and onboarding
- certificate enrollment
- factory test modes left enabled
- service ports and maintenance tools
What to do: Use strong server authentication, consider mutual TLS where appropriate, validate certificate chains correctly, and avoid “accept all certs” fallbacks in production builds.
2) Replay attacks in protocols that lack freshness
Replay attacks reuse valid captured messages. If your protocol does not include nonces, timestamps, sequence numbers, or session binding, an attacker can re-trigger a sensitive action.
What to do: Use session keys, include anti-replay protections, and bind authorization decisions to current session context.
3) Weak randomness and predictable key generation
Some embedded designs still struggle with entropy, especially early boot. Predictable randomness can undermine key generation and session secrets.
What to do: Use a vetted RNG strategy for your platform, avoid rolling your own, and document how entropy is seeded and tested.
4) “Works in the lab” PKI that fails in the field
PKI problems often show up post-launch: expired certificates, no rotation plan, unclear revocation strategy, and brittle provisioning workflows that break manufacturing or servicing.
What to do: Treat certificates like lifecycle assets. Plan rotation, renewal, and revocation from day one, and test failure modes.
Protocols and patterns that work well for connected medical devices
Diffie-Hellman and ECDHE for forward secrecy
Diffie-Hellman style key agreement helps establish session keys without transmitting them directly. Using ephemeral variants (often ECDHE in TLS) supports forward secrecy, meaning past sessions remain protected even if a long-term key is later compromised.
TLS done intentionally (not “default library settings”)
TLS is the common answer for data-in-transit, but configuration matters. Version support, cipher suites, certificate validation behavior, and key sizes should be deliberate and defensible.
Practical framing: In regulated products, “we used TLS” is not enough. You want a short, reviewer-friendly statement of:
- protocol versions supported
- how endpoints authenticate
- how secrets are generated and stored
- how keys and certificates are rotated
- how failures are handled safely
Secure elements and hardware-backed key storage (when warranted)
If compromise of a key materially increases patient safety risk, hardware-backed key protection can be worth it. It can also simplify your threat model story by reducing key extraction likelihood.
What to document for FDA-facing cybersecurity evidence
FDA-facing reviewers typically respond well to structured, traceable evidence. For key exchange, that means showing:
- Threat model coverage: MitM, replay, credential theft, update tampering.
- Security requirements: authentication, cryptography, session management, secure update integrity.
- Architecture views: trust boundaries, certificate flows, key storage boundaries, update signing flow.
- Verification evidence: protocol testing, negative tests, validation of cert handling, downgrade resistance (where applicable).
Blue Goat resources that support the packaging:
- Medical device threat modeling
- FDA-aligned vulnerability and penetration testing
- FDA-compliant SBOM services
- Postmarket cybersecurity management services
A simple checklist: key exchange decisions that should be explicit
- Which trust model do you use (public PKI, private PKI, pinned certs, mutual TLS)?
- How are device identities provisioned at manufacturing?
- Where are private keys stored (filesystem, TEE, secure element, HSM-backed service)?
- How do you rotate and revoke certificates or keys?
- How do you prevent replay and downgrade attacks?
- What happens when keys expire, are lost, or are suspected compromised?
Key takeaways
- In medical device cybersecurity, key exchange is usually the real risk surface, not the encryption algorithm.
- Most failures are implementation and lifecycle failures: authentication gaps, replay exposure, weak randomness, and no rotation plan.
- Treat keys and certificates as lifecycle assets with documented provisioning, rotation, and recovery paths.
- For FDA readiness, focus on traceability: threats → requirements/controls → architecture evidence → verification.
FAQs
Is Diffie-Hellman “enough” to solve key exchange for medical devices?
It solves shared secret establishment, but you still must authenticate endpoints correctly. Without authentication, a MitM can negotiate keys with both sides.
Should a medical device use TLS 1.2 or TLS 1.3?
TLS 1.3 is generally preferred in new designs because it simplifies negotiation and strengthens security properties. If you support TLS 1.2 for interoperability, lock it down with strong suites and validated certificate handling.
Do we need mutual TLS for every device?
Not always. Mutual TLS can reduce impersonation risk, but it adds operational complexity. Make it a risk-based decision and document the rationale.
What is the biggest key management mistake you see in connected devices?
No rotation and no failure mode planning. Keys expire, certificates get revoked, and update signing keys can be compromised. If you do not have a plan, you end up with unsafe workarounds.
How do we test key exchange and TLS beyond “it connects”?
Test downgrade resistance, replay handling, certificate validation edge cases, expired cert behavior, and failure mode safety. Combine automated scanning with targeted negative tests and penetration testing.
Call to action
If you want a clean, FDA-ready package for cryptography and key exchange evidence (threat model, architecture views, verification results, and documentation), we can help.
