Medical Device Cybersecurity Guides
Practical, ungated guides for medical device manufacturers navigating FDA cybersecurity requirements — from premarket submission to postmarket operations.

The MedTech Cybersecurity Standards Decoder
FDA Section 524B, IEC 81001-5-1, AAMI SW96, ISO 14971, and more — what each standard requires, how they connect, and exactly what FDA expects to see in your submission.
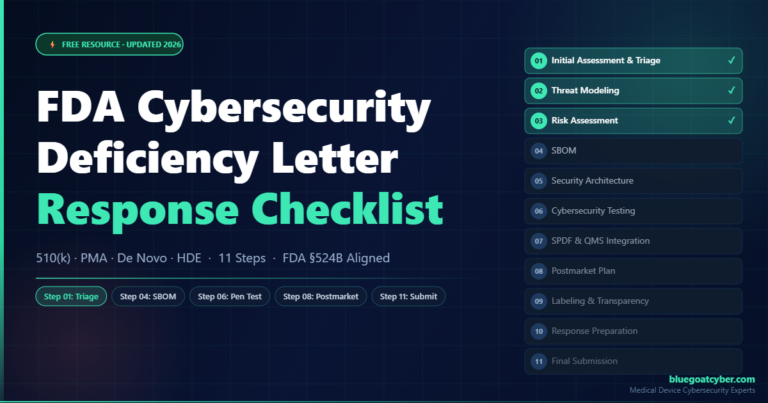
FDA Cybersecurity Deficiency Letter Response Checklist
A step-by-step 11-stage checklist for organizing and resolving an FDA cybersecurity deficiency letter for 510(k), PMA, De Novo, and HDE submissions.

10 Reasons Medical Device Cybersecurity Vendors Fail You
Before you hire a medical device cybersecurity vendor, read this. The 10 most common ways vendors fail manufacturers — and how to spot them early.

12 Critical Findings from Medical Device Penetration Tests
What real medical device penetration tests actually find — the 12 most critical and recurring vulnerabilities, with remediation guidance for each.

12 Reasons the FDA Rejects Medical Device Cybersecurity Submissions
The 12 most common reasons FDA sends back cybersecurity submissions, explained with specifics on what reviewers expect instead.
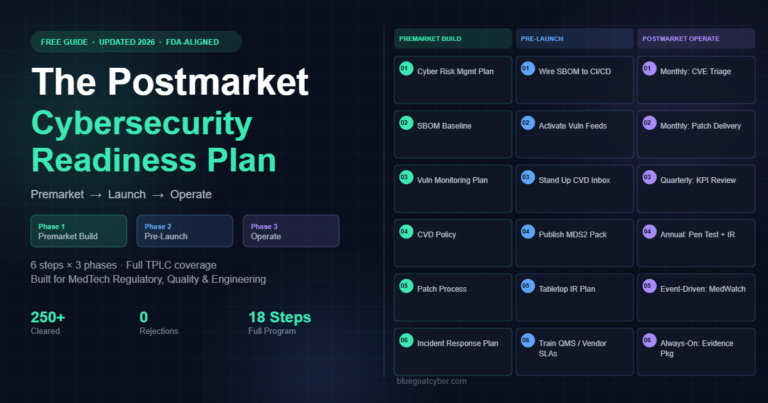
The Postmarket Cybersecurity Readiness Plan
A three-phase, 18-step operational plan for maintaining FDA cybersecurity compliance across your device’s full postmarket lifecycle.

The SPDF Playbook for FDA-Ready Medical Devices
The 8-pillar Secure Product Development Framework that FDA accepts — what each pillar produces, how they connect, and a 15-point pre-submission readiness checklist.

12 Critical Threat Modeling Gaps in Medical Device Submissions
A practical self-assessment guide to the 12 threat modeling gaps that trigger FDA cybersecurity questions, with specific fixes for each.
Put the Guides Into Action
Book a free discovery session. We respond within 24 hours with a quote — no sales pressure, just a clear assessment and fixed-price scope.
