When teams talk about “secure operating systems” for medical devices, microkernels come up fast. The idea sounds simple: keep the kernel small, move services (like drivers, filesystems, and networking) out of the kernel, and gain better isolation. That can be a real advantage in safety-critical products, but it is not automatic security. The details matter.
This article explains microkernels in practical terms for MedTech teams and gives you a checklist you can use to design, test, and document a microkernel-based architecture in an FDA-friendly way.
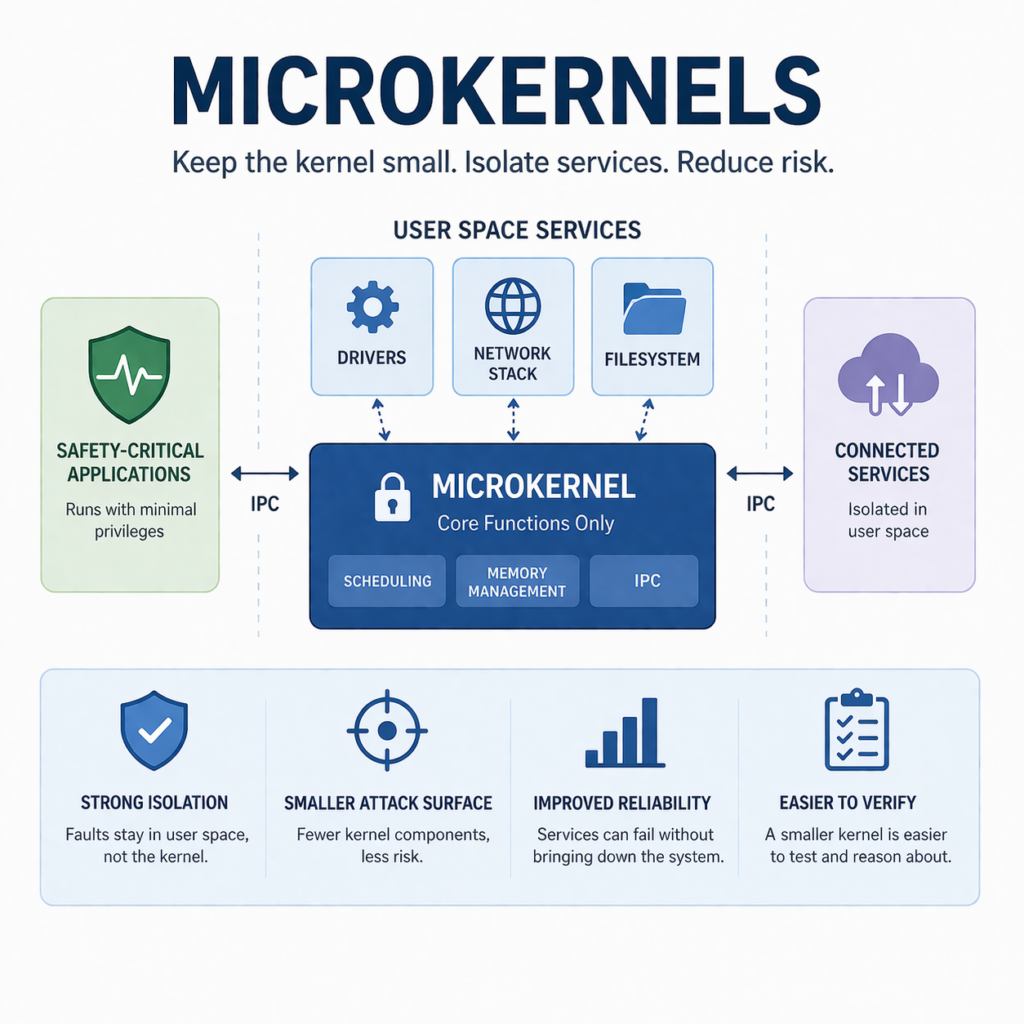
What is a microkernel?
A microkernel is a kernel design that keeps only a small set of core functions in privileged kernel space. Those core functions usually include scheduling, memory management, and inter-process communication (IPC). Other services that would typically live inside a monolithic kernel run in user space as separate processes.
In a microkernel architecture, the OS looks more like a set of cooperating components than one large block of privileged code. The kernel coordinates, user-space services provide functionality, and IPC is the glue.
Microkernel vs monolithic vs “hybrid” kernels
Monolithic kernel (traditional)
Most services run in kernel space. This can be fast and straightforward, but it also means a bug in a driver or subsystem can have a high impact because it runs with full kernel privileges.
Microkernel
Only essential mechanisms stay in the kernel. Many services run in user space with tighter privilege and stronger fault isolation. This can shrink the trusted computing base, but you also introduce IPC design complexity and performance considerations.
Hybrid approaches
Many real-world systems take a blended approach. What matters most is not the label, but where you draw privilege boundaries and how you enforce them.
Why microkernels matter for medical device cybersecurity
In a connected medical device, “cybersecurity” is rarely a single control. It is how you limit the blast radius when something goes wrong. Microkernels can support that goal when you use them to create real isolation between:
- Safety-critical functions (therapy delivery, alarms, essential performance)
- High-risk interfaces (networking, file parsing, removable media, remote service channels)
- Support services (logging, UI, telemetry, update clients)
When the isolation is real, a bug in a non-safety service is less likely to become a full device compromise. That is the value proposition.
FDA’s current premarket guidance emphasizes secure-by-design, architecture clarity, and verification evidence across the lifecycle. If your OS choice is part of your security argument, you should be ready to show how it supports isolation, least privilege, updateability, and vulnerability response. See the FDA guidance here: FDA: Cybersecurity in Medical Devices (Premarket Guidance).
Where microkernels show up in safety-critical systems
Microkernel ideas have a long history in safety-critical domains. A few commonly cited examples include seL4 (known for formal verification work) and QNX Neutrino (known for message-passing architecture in embedded systems). If you want to go deeper on these reference implementations:
These are examples, not recommendations. The right OS for your device depends on your architecture, safety case, maintenance plan, and ability to produce evidence.
The tradeoffs MedTech teams need to understand
1) Performance and latency
Microkernels rely heavily on IPC. That can introduce overhead, especially if services are chatty or poorly partitioned. In many embedded systems the overhead is manageable, but you still need to measure it and make sure it does not affect essential performance.
2) Complexity moves, it does not disappear
A microkernel reduces kernel complexity, but pushes more functionality into user-space services. That can be great for isolation, but it creates more components, more interfaces, and more integration testing.
3) Driver ecosystem and hardware support
Device drivers are often where risk lives. Microkernels can help by running drivers in user space, but only if your platform and vendor ecosystem support that cleanly.
4) Security depends on policy, not architecture diagrams
“We use a microkernel” is not a control. The controls are the actual enforcement mechanisms: capability or permission models, memory protection, IPC authorization, secure boot, signed updates, logging, and response procedures.
A practical microkernel security checklist for medical devices
Design
- Partition intentionally: identify safety-critical functions and isolate them from networking and parsing surfaces.
- Define trust boundaries: document which components are trusted, which are exposed, and what happens if each fails.
- Least privilege: ensure each service runs with only the permissions it needs.
- Harden IPC: treat IPC endpoints like APIs. Authenticate and authorize requests between components where applicable.
Build and configure
- Secure boot and secure update: make it hard to persist in the device even if a service is compromised.
- Memory protections: turn on platform hardening (ASLR/DEP or equivalents) where supported.
- Logging that is usable: log security-relevant events, privilege failures, and update events with enough fidelity to investigate issues.
Test and prove it
- Negative tests: prove components cannot access resources they should not access.
- Fault injection: crash or degrade non-safety services and show the safety-critical path stays safe.
- Interface testing: fuzz or stress parsers and IPC endpoints for robustness.
- Security testing: validate exploitability assumptions, not just functional behavior.
If you want support building this into your design process, Secure MedTech Product Design Consulting and Medical Device Threat Modeling Services are designed for exactly this kind of architecture and evidence work.
What to document for FDA-facing credibility
FDA reviewers typically respond well to clear, testable architecture narratives. For a microkernel-based design, your documentation set should make these points obvious:
- Architecture views: partitions, privilege boundaries, IPC flows, update paths, and trust boundaries.
- Risk management linkage: threats tied to components and interfaces, plus mitigations and residual risk rationale.
- Third-party software story: what you run, how you track it, and how you keep it updated, ideally supported by an SBOM.
- Verification evidence: tests that prove isolation, least privilege, and failure containment.
- Postmarket plan: monitoring, patch delivery, and vulnerability response over the lifecycle.
Helpful internal resources:
- FDA Premarket Cybersecurity Services
- FDA-Compliant SBOM Services for MedTech
- Medical Device Cybersecurity Penetration Testing
- FDA Postmarket Cybersecurity Services
- FDA Medical Device Cybersecurity 2026: 524B eSTAR Checklist
Key takeaways
- Microkernels can improve isolation by keeping the kernel small and moving services to user space.
- The security benefit comes from real privilege boundaries, not from the word “microkernel.”
- Tradeoffs include IPC design complexity, performance considerations, and driver ecosystem constraints.
- For FDA-facing evidence, focus on architecture clarity, risk traceability, and verification results that prove isolation and failure containment.
FAQs
Are microkernels “more secure” than monolithic kernels?
They can be, because a smaller trusted computing base and stronger isolation can reduce blast radius. But the real outcome depends on how you partition services, enforce privilege, harden IPC, and maintain the system over time.
Does the FDA require a microkernel for connected medical devices?
No. FDA expects reasonable assurance of cybersecurity based on risk. You can meet that expectation with different architectures if you can document and verify your controls.
Do microkernels eliminate the need for secure coding?
No. You still need secure coding, testing, and vulnerability management. A microkernel can limit impact of a bug, but it does not prevent bugs.
How do we prove isolation in testing?
Use negative tests for privilege boundaries, fault injection to show safety-critical paths remain safe, and targeted security testing for exposed interfaces and IPC flows.
What is the biggest mistake teams make when adopting a microkernel?
Assuming the architecture alone provides security. The hard work is in the permission model, IPC controls, update and logging design, and evidence that those controls work.
Book a Discovery Session
If you want help deciding whether a microkernel approach supports your device’s safety and cybersecurity goals, and you want documentation and test evidence that reviewers can follow, we can help.
Conclusion
Microkernels are a useful tool for safety-critical systems because they can make isolation more practical. In MedTech, the smartest way to use that advantage is to design around trust boundaries, enforce least privilege, test failure containment, and document the results. That combination tends to stand up well in both engineering reality and regulatory review.
