Cybersecurity guides for MedTech teams.
Practical playbooks, checklists and decoders we use on every engagement.
43 guides
MedTech Cybersecurity Primer for Busy Founders
250+ 0 6–10 wk FDA submissions supported Cybersecurity rejections Class II eSTAR cyber pack SINCE 2014 TRACK RECORD TYPICAL TIMELINE
Read the guideMedTech Cybersecurity Vendor Evaluation Grid
MedTech Cyber Vendor Evaluation Grid A scoring grid for cybersecurity firms tailored to MedTech regulatory needs.
Read the guideMSA Redlines Playbook for MedTech Cyber Vendors
MSA Redlines: Usually Accepted vs. Not A short playbook for the redlines we see most often, what we accept without negotiation, and what we push back on and why.
Read the guidePatient-Safety-First Threat Modeling Guide
Patient-Safety Threat Modeling Worksheet A right-sized STRIDE pass that maps cybersecurity threats to ISO 14971 hazards.
Read the guidePenetration Test Refresh Guide
Pen Test Refresh vs. Full Re-Test Decision Guide When a delta-only refresh is sufficient, and when FDA expects a full re-test.
Read the guide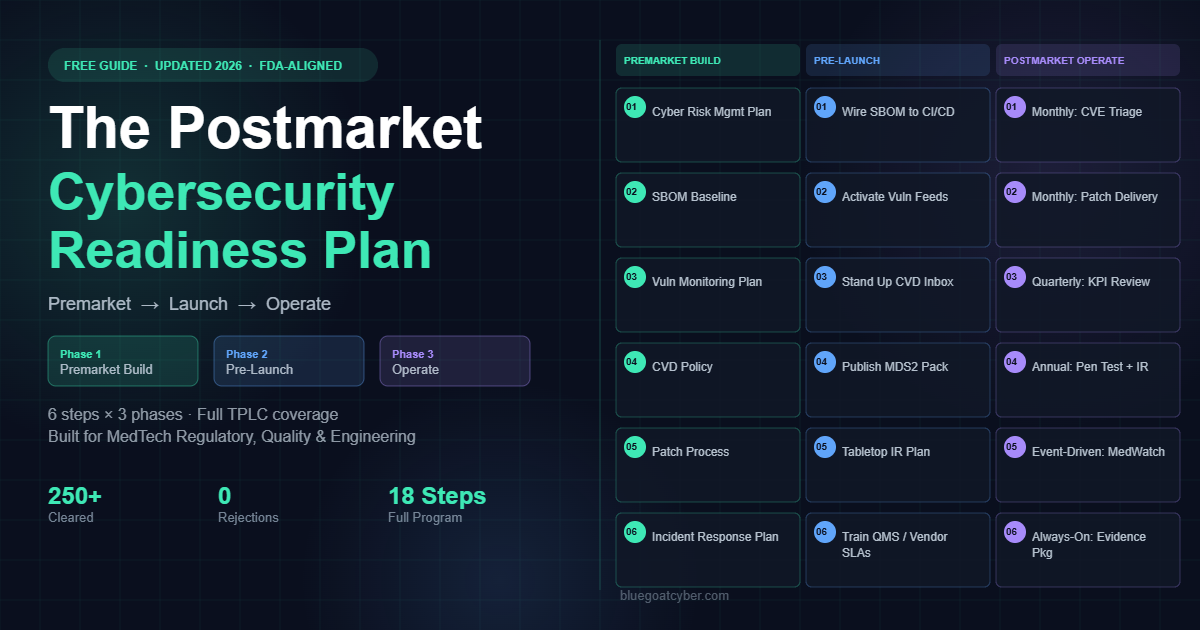
Postmarket Cybersecurity Readiness Plan
What you need in place after clearance to satisfy FDA postmarket expectations and stay ahead of vulnerabilities.
Read the guideRe-Engagement Sequence: Restarting a Stalled Cyber Program
Re-Engagement Sequence (After Going Dark) A plain, no-pressure sequence to re-open a cybersecurity conversation after a quiet period.
Read the guideRight-Sized Threat Modeling for MedTech
250+ 0 6–10 wk FDA submissions supported Cybersecurity rejections Class II eSTAR cyber pack SINCE 2014 TRACK RECORD TYPICAL TIMELINE
Read the guideSection 524B Post-Market Retrofit Guide
250+ 0 6–10 wk FDA submissions supported Cybersecurity rejections Class II eSTAR cyber pack SINCE 2014 TRACK RECORD TYPICAL TIMELINE
Read the guideBring this rigor to your next submission.
Book a 30-minute strategy session and we'll map the guides to your actual device, timeline and gaps.
