
From Idea to FDA Clearance: What Nobody Tells MedTech Founders
The biggest MedTech startup mistake isn’t the tech. Learn why clarity, FDA planning, quality systems, and cybersecurity determine success.
Read articleEvery article in our archive in FDA.
Showing 12 of 46 articles in FDA · Page 2 of 4

The biggest MedTech startup mistake isn’t the tech. Learn why clarity, FDA planning, quality systems, and cybersecurity determine success.
Read article
FDA’s QMSR is now in effect. See why connected MedTech teams must build cybersecurity into the QMS - risk management, V&V, suppliers, and postmarket.
Read article
Learn the difference between measures and metrics, FDA expectations, and how medical device makers use them to improve cybersecurity and protect patients.
Read article
Learn how to organize FDA medical device cybersecurity requirements into 18 key deliverables, from threat modeling and SBOMs to testing and labeling.
Read article
Proactive medical device cybersecurity drives faster FDA clearance, investor trust, and patient safety.
Read article
Manufacturers in any industry often seek regulations as barriers or obstacles. They may add costs to production or layers of oversight. Those are usually not palatable to business, but in medical device cybersecurity, these rules are much more than red tape. The Food & Drug Administration (FDA) rece
Read article
Cybersecurity is no longer optional - it’s critical to patient safety and regulatory success. As medical devices increasingly connect to apps, cloud platforms, and hospital networks, manufacturers must follow stringent cybersecurity practices throughout the entire product lifecycle. At Blue Goat Cyber
Read article
Navigating the FDA clearance process for medical devices involves more than technical documentation and testing - it involves significant regulatory costs that manufacturers must plan for early. Whether you're submitting a 510(k), Premarket Approval (PMA), or a De Novo request, understanding the latest
Read article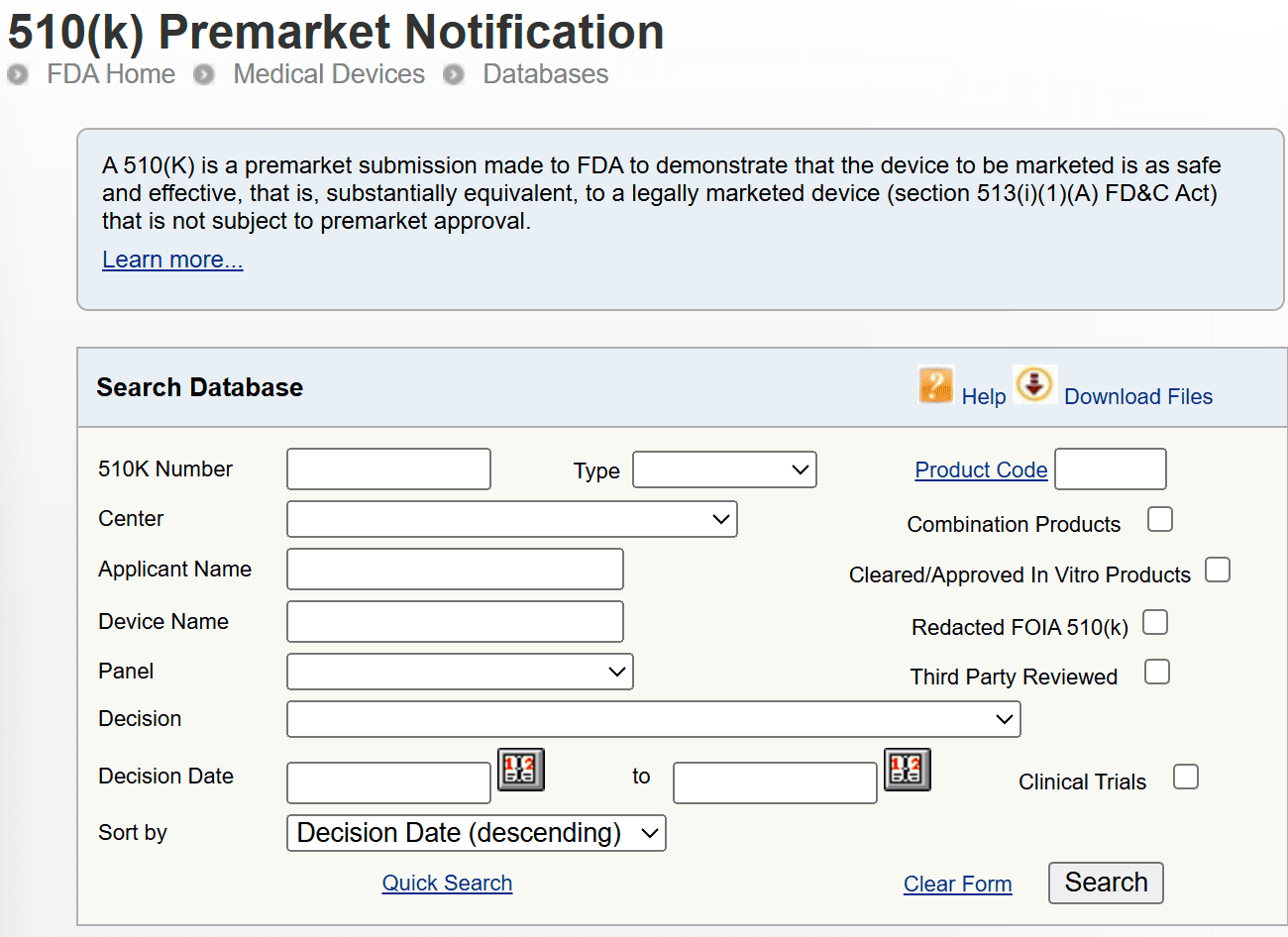
Updated April 12, 2025 Understanding how to mine FDA databases for insights is a strategic advantage if you're bringing a medical device to market or managing one post-approval. These databases aren’t just regulatory archives. They're treasure troves of competitive intelligence, predicate data, and
Read article
How verification, validation, and regression testing work together to produce defensible FDA premarket cybersecurity evidence under Section 524B, IEC 62304, and the SPDF.
Read article
How to secure real-time operating systems (FreeRTOS, VxWorks, QNX, Zephyr) inside medical devices and evidence the controls in a §524B premarket cybersecurity submission.
Read article
Discover the ins and outs of a Modular PMA Submission, a streamlined approach to gaining FDA clearance for medical devices.
Read article30-minute strategy session. No cost, no commitment - just answers from people who've shipped 250+ submissions.