IVD Cybersecurity Risks
Uncover the potential dangers lurking within in vitro diagnostic (IVD) medical devices in this eye-opening article.
Read articleEvery article in our archive tagged Primer.
Showing 12 of 127 articles tagged Primer · Page 6 of 11

Uncover the potential dangers lurking within in vitro diagnostic (IVD) medical devices in this eye-opening article.
Read article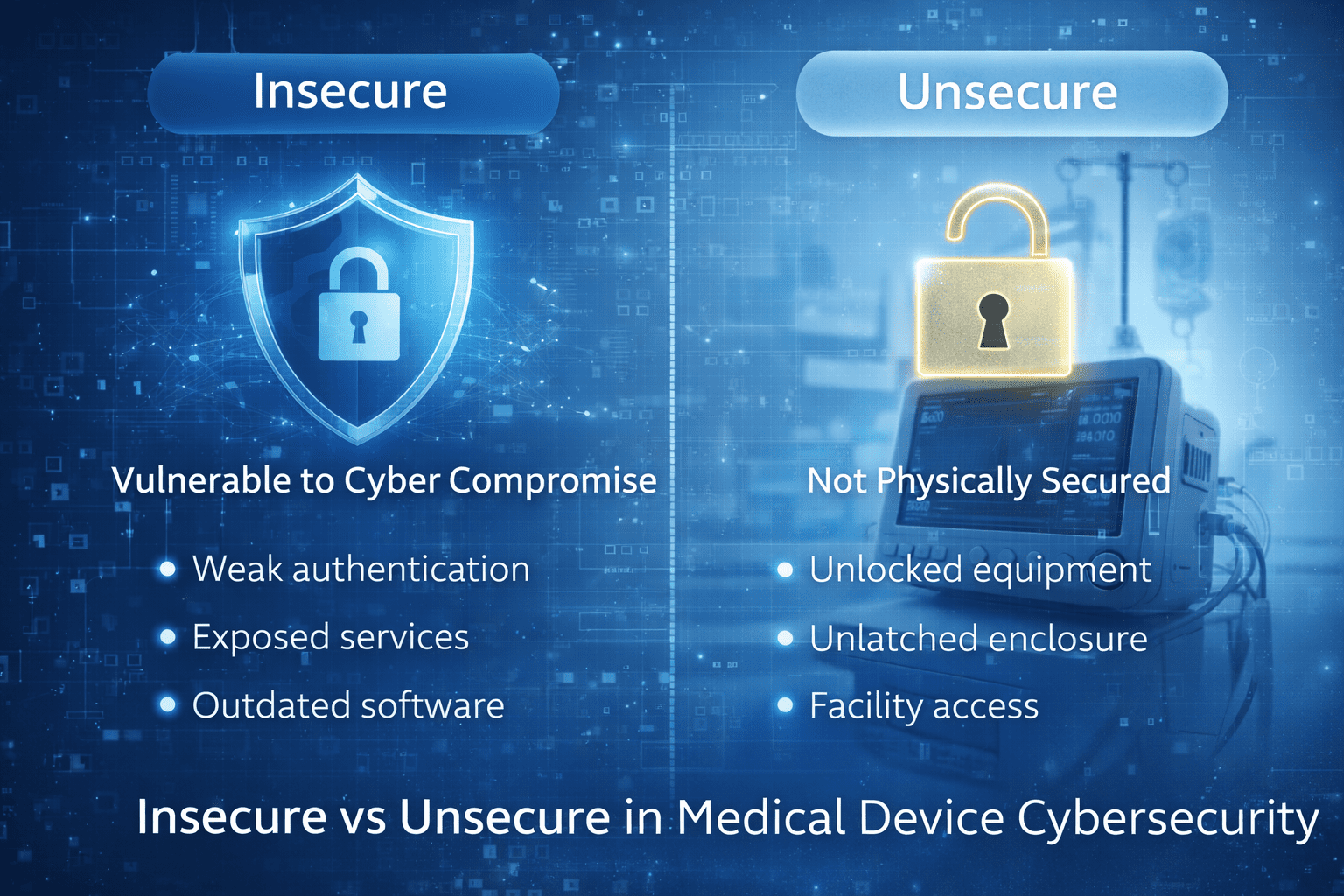
Insecure vs unsecure isn’t just grammar. Learn the difference and why precise wording matters for medical device cybersecurity docs and labeling. For FDA teams.
Read article
The “hoodie hacker” cliché distorts risk, weakens hiring, and misguides security decisions. Here’s what MedTech teams should do instead.
Read article
Digital Imaging and Communications in Medicine, or DICOM, is the industry standard for medical imaging formats. This format has expanded functionality compared to typical image formats, as it can store patient data and procedure information along with the actual image. Typically, a specialized DICOM
Read article
Discover the ins and outs of DICOM vulnerabilities in the MedTech industry with this comprehensive overview.
Read article
Can contact lenses fool iris scans? Learn how iris spoofing affects medical device cybersecurity, liveness detection, and FDA lifecycle expectations.
Read article
Learn how to identify and address vulnerabilities in PACS medical devices with our comprehensive guide.
Read article
Medical device cybersecurity doesn't require genius hackers - just neglected vulnerabilities. Discover how Blue Goat protects devices, patients, and compliance.
Read article
Ephemeral ports affect firewall rules, logging, and threat models for connected medical devices. Learn ranges, risks, and practical controls.
Read article
Discover the potential risks and implications of LoRaWan vulnerabilities on the cybersecurity of medical devices.
Read article
Discover the crucial role of RFID technology in safeguarding medical devices from cyber threats.
Read article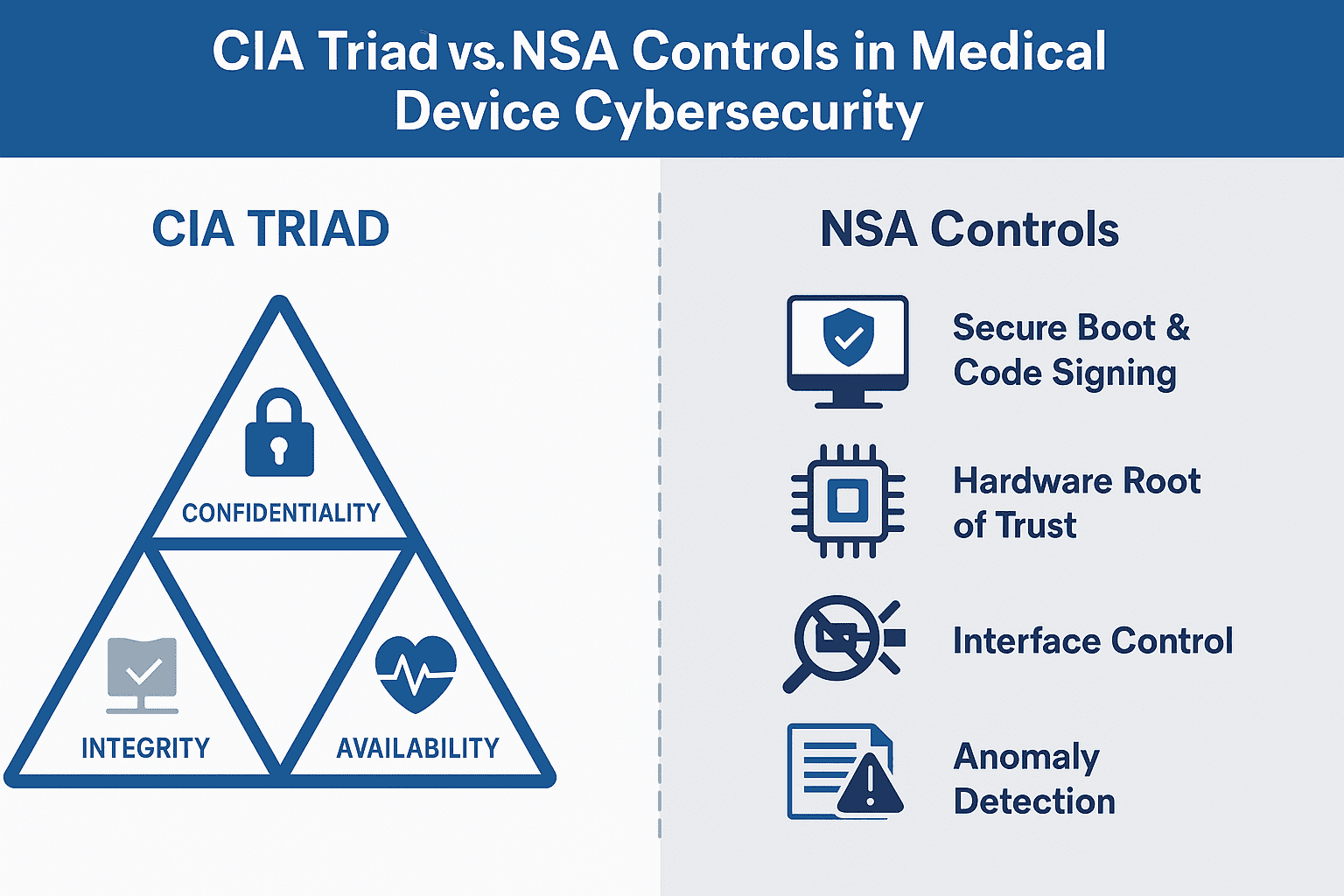
Explore how the CIA Triad and NSA cybersecurity controls strengthen medical device security and support FDA compliance from design to postmarket.
Read article30-minute strategy session. No cost, no commitment - just answers from people who've shipped 250+ submissions.