
IPC Vulnerabilities in Medical Devices: Risks and Controls
Learn how IPC weaknesses enable privilege escalation and unsafe device behavior - and how to design, test, and document mitigations for FDA.
Read articleEvery article in our archive tagged Risk.
Showing 12 of 47 articles tagged Risk · Page 3 of 4

Learn how IPC weaknesses enable privilege escalation and unsafe device behavior - and how to design, test, and document mitigations for FDA.
Read article
Learn why medical device hazard analysis and critical control points are crucial for ensuring the safety and effectiveness of medical devices.
Read article
Uncover the potential dangers lurking within in vitro diagnostic (IVD) medical devices in this eye-opening article.
Read article
Explore the crucial disparities between unsigned and signed integers in this comprehensive article.
Read article
Delve into the fascinating world of self-aware AI as we explore the concept, implications, and potential of artificial intelligence that possesses self-awareness.
Read article
Discover the fascinating world of Bluetooth technology with this comprehensive guide to the different types of Bluetooth.
Read article
The origin of “2600” has a modern lesson: how to rank medical device cyber risk, prioritize fixes, and document decisions for regulated products.
Read article
NFC security for medical devices: common threats like eavesdropping, relay, and tag tampering - plus practical mitigations for design, testing, and evidence.
Read article
Understand WPA2’s 4-way handshake, real risks like KRACK and weak passphrases, and practical mitigations for connected medical device Wi-Fi ecosystems.
Read article
VM escape breaks VM isolation to reach the host or hypervisor. Learn MedTech scenarios, controls, and evidence for FDA-aligned security.
Read article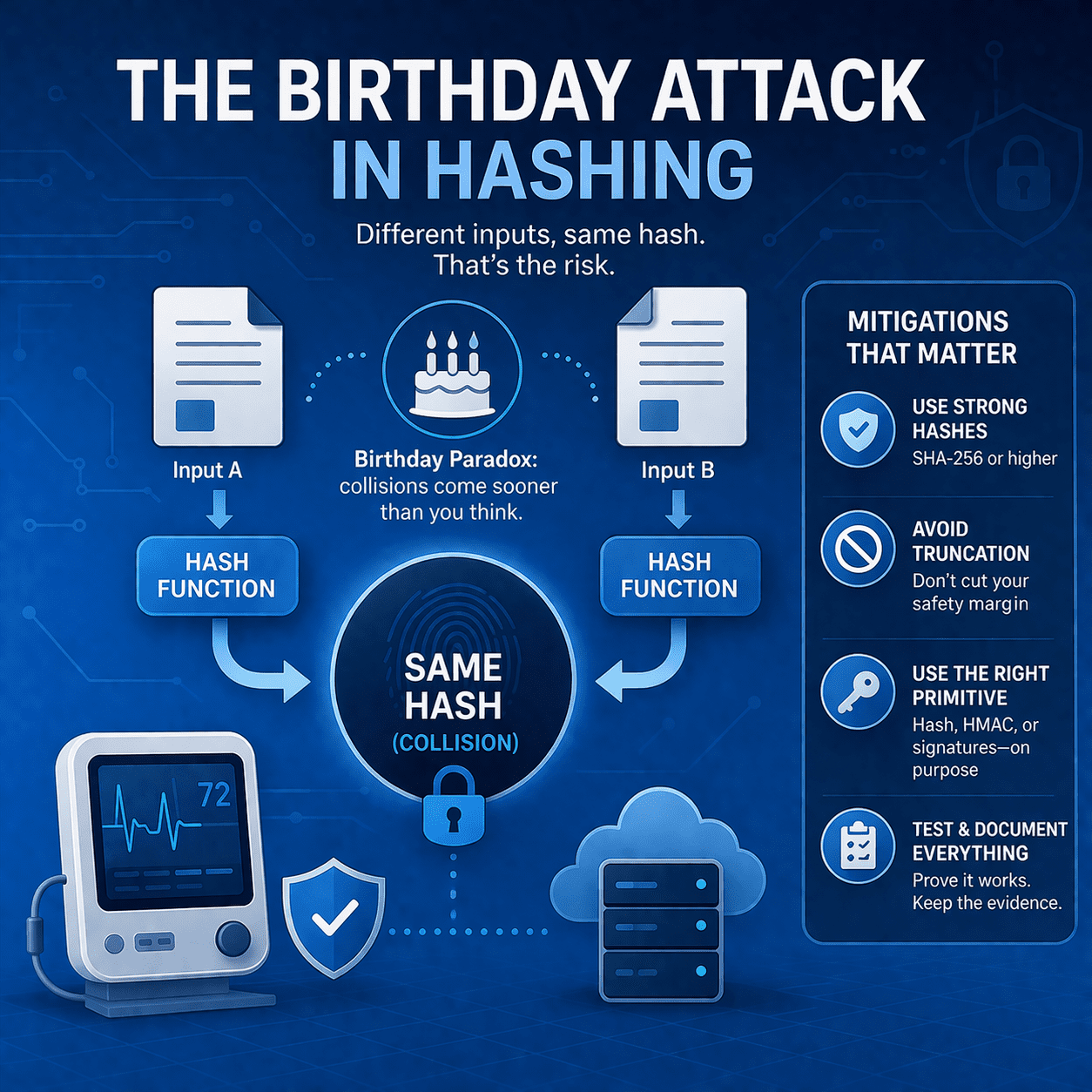
Learn how birthday attacks exploit hash collision probability - and what MedTech teams should do (SHA-256+, signing, truncation rules, testing, evidence).
Read article
Learn how Shodan exposes connected medical devices - and how manufacturers and hospitals can use it to identify risks, improve security, and meet FDA expectations.
Read article30-minute strategy session. No cost, no commitment - just answers from people who've shipped 250+ submissions.