Mitigating Interoperable Medical Device Risk
Discover how to navigate the complexities of interoperable medical devices and effectively manage associated risks.
Read articleEvery article in our archive tagged Risk.
Showing 11 of 47 articles tagged Risk · Page 4 of 4

Discover how to navigate the complexities of interoperable medical devices and effectively manage associated risks.
Read article
Discover how medical devices are meticulously designed with patient safety as the top priority.
Read article
Learn how ASLR reduces buffer overflow impact, where it falls short, and practical controls MedTech teams can use to prevent memory corruption in devices.
Read article
Heap spraying increases exploit reliability for memory bugs. Learn MedTech defenses: patching, mitigations, secure coding, fuzzing, and FDA-ready evidence.
Read article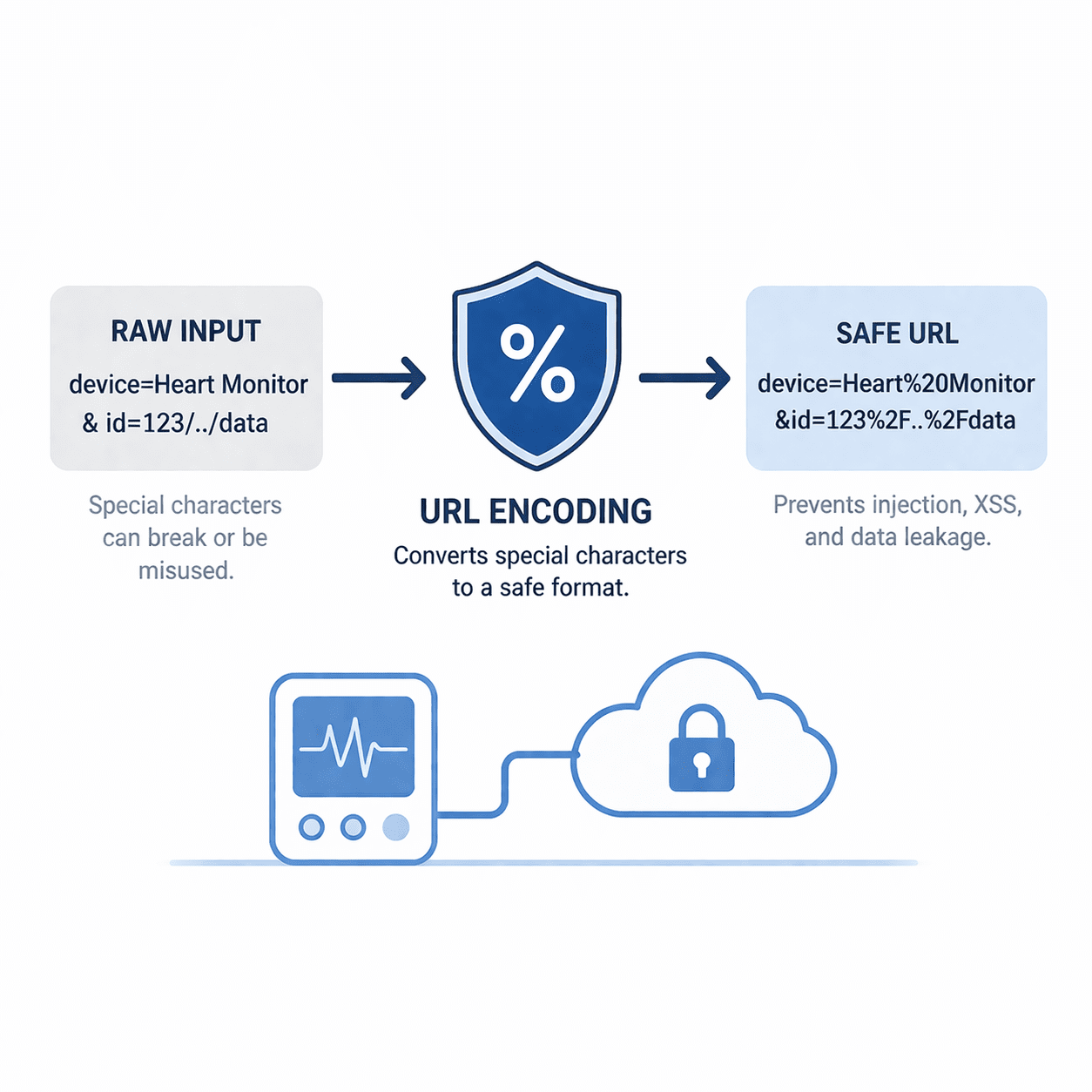
URL encoding prevents parsing mistakes that lead to injection, XSS, and data leakage in MedTech portals and APIs. Includes a practical checklist.
Read article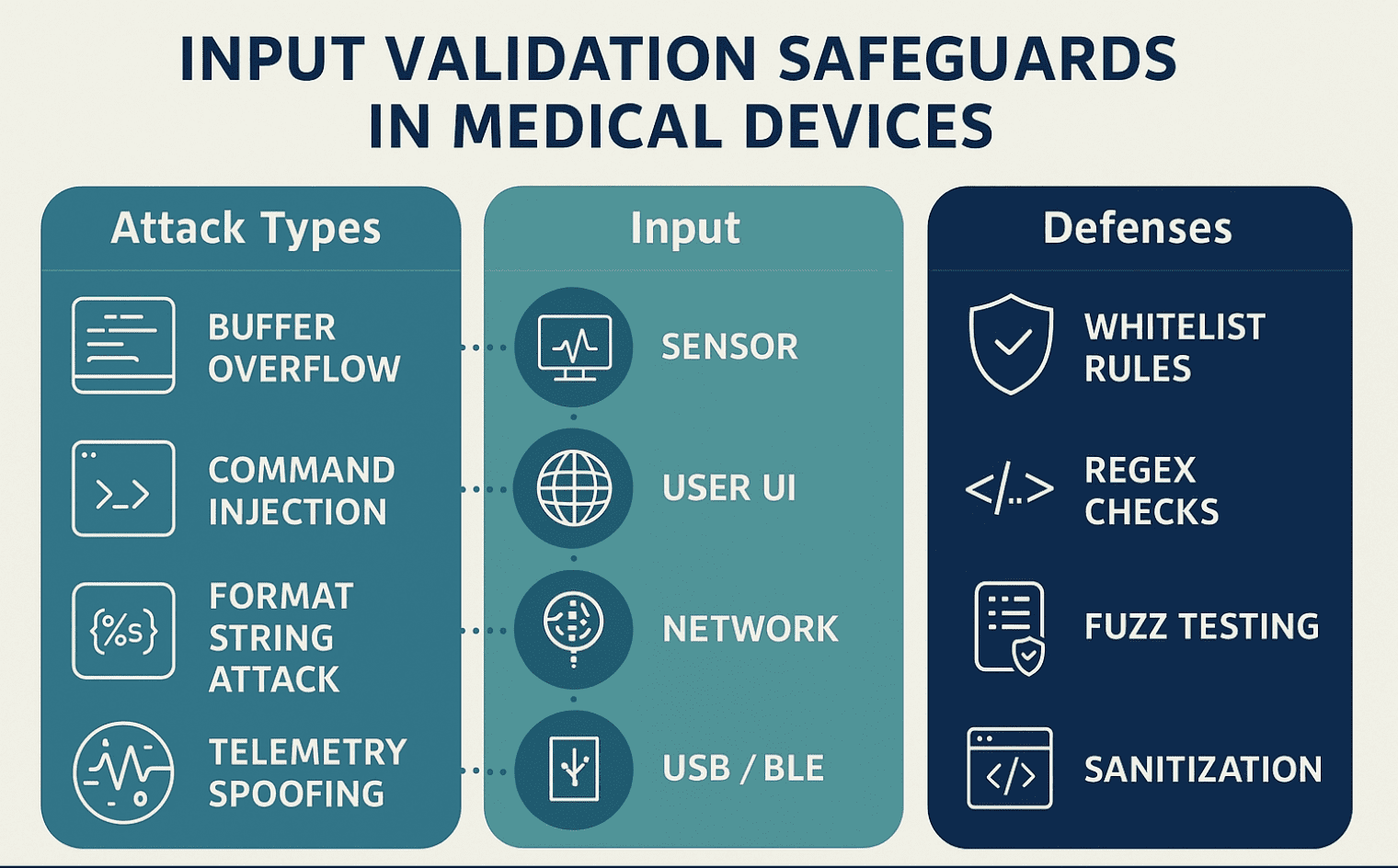
Learn how input validation in medical devices prevents buffer overflows, command injections, and supports FDA cybersecurity compliance.
Read article
Explore the intricacies of risk-based testing for medical device software in this comprehensive guide.
Read article
A practical guide to AAMI TIR57 (R2023) and how it supports FDA’s Feb 2026 cybersecurity guidance - risk analysis, controls, and evidence.
Read article
In this post, we unravel this complex issue of cyber threats in medical devices and discuss what manufacturers can do to make their devices more secure.
Read article
Learn how ISO 14971 risk management applies to medical device cybersecurity - identify cyber hazards, control residual risk, and align with FDA expectations.
Read article
This blog lists the 50 cybersecurity issues in medical devices and explains how penetration testing could have prevented them.
Read article30-minute strategy session. No cost, no commitment - just answers from people who've shipped 250+ submissions.