SoC Vulnerabilities in Medical Devices: What Manufacturers Need to Know
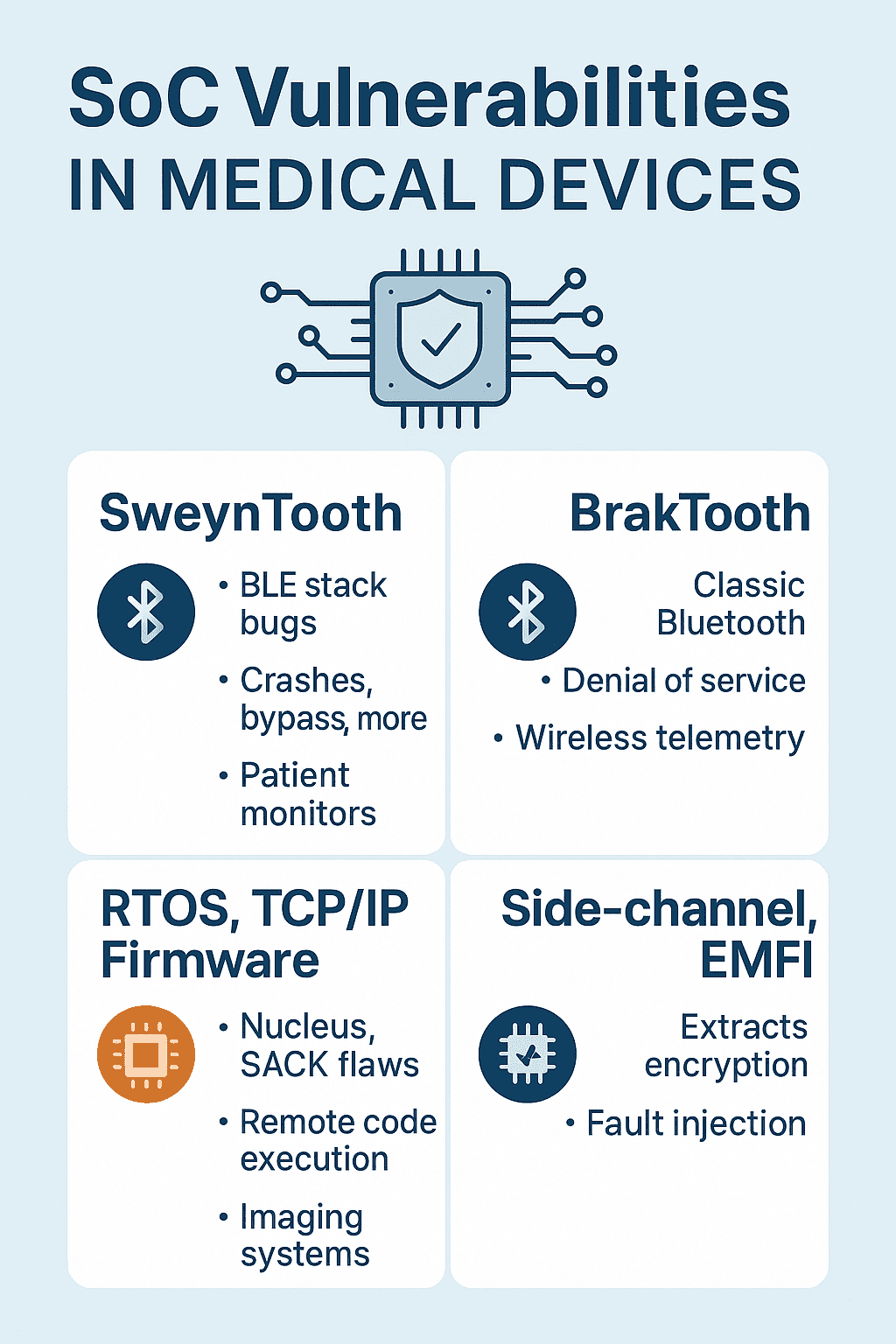
Learn how SoC vulnerabilities (SweynTooth, BrakTooth, NUCLEAUS) threaten connected medical devices - and what manufacturers need to secure and comply.
Read articleDeep dives on FDA expectations, threat modeling, penetration testing, SDLC, and the standards your team is being asked to meet.
Showing 12 of 360 articles · Page 18 of 30

Learn how SoC vulnerabilities (SweynTooth, BrakTooth, NUCLEAUS) threaten connected medical devices - and what manufacturers need to secure and comply.
Read article
Key escrow in medical device cybersecurity: when it’s appropriate, risks it introduces, and how to align cryptographic key management with FDA expectations.
Read article
Updated November 16, 2024 The cybersecurity of medical devices has emerged as a critical concern for manufacturers, healthcare providers, and regulatory bodies. The Manufacturer Disclosure Statement for Medical Device Security (MDS²) plays a pivotal role in addressing these concerns by providing a s
Read article
Updated November 16, 2024 The cybersecurity of medical devices has emerged as a critical concern for manufacturers, healthcare providers, and regulatory bodies. The Manufacturer Disclosure Statement for Medical Device Security (MDS²) plays a pivotal role in addressing these concerns by providing a s
Read article
Learn FMEA for medical devices with practical examples - including cybersecurity failure modes - so QA/RA and engineering teams can prioritize risk and actions.
Read article
Learn how IPC weaknesses enable privilege escalation and unsafe device behavior - and how to design, test, and document mitigations for FDA.
Read article
Learn why medical device hazard analysis and critical control points are crucial for ensuring the safety and effectiveness of medical devices.
Read article
Learn how to safeguard your implantable medical devices from cyber threats with these essential cybersecurity measures.
Read article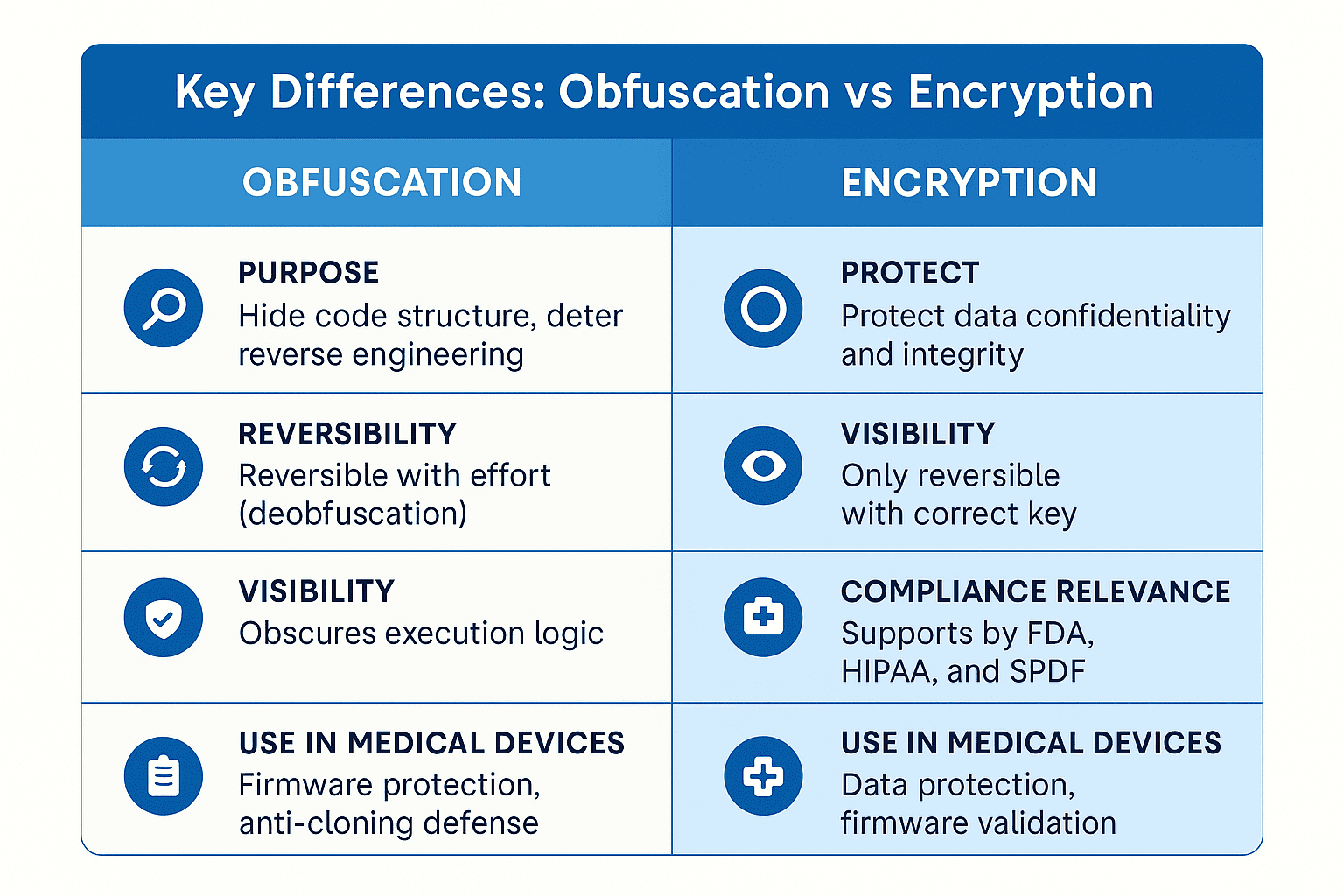
Obfuscation and encryption protect medical devices in different ways. Learn when to use each to secure data and firmware and meet FDA cybersecurity guidance.
Read article
The Bluetooth Low Energy (BLE) vulnerabilities that matter most for FDA-regulated medical devices, and how to evidence them in a §524B premarket cybersecurity submission.
Read article
Learn why implementing a robust Medical Device Quality Management System (QMS) is crucial for ensuring product safety, regulatory compliance, and overall efficiency in the healthcare industry.
Read article
Discover the critical role of cybersecurity in safeguarding surgical robots from potential threats.
Read article30-minute strategy session. No cost, no commitment - just answers from people who've shipped 250+ submissions.